Aromatase is a cytochrome p450 enzyme that converts male hormones (testosterone and androstenedione) to female hormones (estradiol and estrone, respectively). In women, the estradiol produced by this reaction contributes to the proliferation of the epithelial cells in the breast. Accordingly, oncologists prescribe aromatase inhibitors to breast cancer patients to attenuate tumor progression.
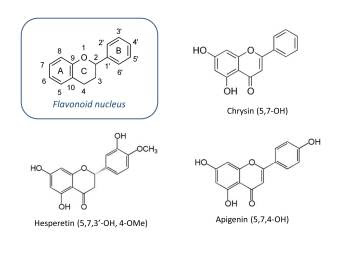
For an illustrious node in steroid biochemistry and desperado of preventive nutritional endocrinology, aromatase is submissive and curiously apathetic to a cavalier lot of structurally diverse inhibitors. From alkaloids to peptides to polyphenols, aromatase will acquiesce with an oblique glance of regret at most. Hence, naturally occurring aromatase inhibitors in the plant kingdom number in the hundreds. Polyphenols, particularly those with the flavonoid nucleus are the most effective. Specifically, hydroxyflavones, which feature a 4-keto group and more than one hydroxyl group, inhibit aromatase with potency that is respectable for natural products (IC50 values in the low micromolar range).
Among the flavones, chrysin and apigenin are the most potent. Since their inhibitory performance is much weaker than today’s drugs, high concentrations are required in vivo. Poor bioavailability of these compounds is partially attributed to phase II conjugation of the aromatic hydroxyl groups. Although methylation at these positions might improve metabolic stability, such modification concomitantly diminishes aromatase inhibitory potency. Flavonoid-aromatasestructure-activity relationships are well established in vitro, but to arrive at the best candidate for nutraceutical product development, a compromise must be made.
The orange: A potential answer, and many questions
Citrus fruits intersect female reproductive physiology through multiple modes of action, and aromatase is one of many putative targets. Hesperetin is the aglycone of hesperidin, a flavone found in oranges. Compared to most polyphenols, it is a crumby antioxidant (this is actually a good thing when it comes to aromatase inhibition). Hesperetin is structurally similar to the most potent dietary inhibitors, apigenin and chrysin, and some studies report similar IC50 values[1]. But the reason I am so fascinated is simple— bioavailability.
In a recent clinical study in which volunteers drank 2-3 glasses of orange juice, hesperetin accumulated in plasma to a peak level of 5 uM, which is within the range of aromatase inhibition in vitro. In a pharmacokinetic study of a micronized hesperetin preparation, a Cmax of 10 uM was reported following a single dose of 150 mg [2]. Compared to nanomolar concentrations observed for many flavonoids at doses exceeding 100 mg, these numbers are respectable. However, clinically meaningful inhibition requires that the flavone reach the target tissue, e.g. the breast. In a recent study, hesperetin reduced the growth of aromatase-expressing tumors in mice, supporting adequate distribution to mammary tissue [3].
Clinical trials in humans have yet to examine hesperetin as a viable tool in mitigating estrogen production in breast cancer. Until further scientific progress is made, I will have to cope with two burning questions. First, does the hydroxyflavone structure, which is reminiscent of soy phytoestrogens, allow hesperetin to bind the estrogen receptor, comprising a potentially cooperative or functionally antagonistic mechanism? The second question is simpler, but so much harder to answer–is protection of the cells that uphold the 2nd leading cause of cancer deaths in women too much to ask of a sweet, unassuming, deeply habitual Valencia?

