Quercetin is an immunomodulator that belongs to a class of low-molecular weight polyphenols known as flavonoids. The subject of over 10,000 published studies and scholarly reviews to date, it’s the most extensively characterized flavonol in the human diet. By virtue of its ability to donate electrons, quercetin is best known for its antioxidant properties, which are superior to most other flavonoids, including luteolin, rutin and hesperidin [1, 2]. Its clinical utility in immunology stems from a far more extensive pharmacodynamic profile involving anti-inflammatory and mast cell stabilizing properties [1-5].
Why does quercetin have poor oral bioavailability?
The aromatic rings and hydroxyl groups that endow quercetin with antioxidant and immunomodulating properties also impair its ability to cross membranes and resist rapid metabolism [2]. Pharmacokinetic studies indicate that only 20-30% of an oral dose is absorbed [6,7], which is due to the following factors:
- Low aqueous solubility limits dissolution in the intestine. Phytosomes and glucose conjugation are two ways to solve this.
- Poor passive diffusion makes membrane permeation slow and incomplete. Phytosomes improve passive diffusion. Alternatively, glucose conjugation assigns the compound to use a carrier instead.
- Rapid presystemic phase II metabolism (mainly glucuronidation) impairs bioavailability by adding large, polar adducts that accelerate biliary and urinary excretion. Phase II inhibitors can attenuate this process.
Solving for #1 and 2 (Low aqueous solubility and poor passive diffusion)
Phytosomes have proven efficacy for enhancing solubility, membrane permeation and intestinal absorption of polyphenols, including curcumin and quercetin. QuerceFit® phytosome technology combines quercetin and phospholipids in a 1:1 ratio, promoting resistance to degradation in the digestive tract and facilitating passive diffusion across the intestinal membranes. In a randomized crossover human pharmacokinetic study, the phytosome enhanced bioavailability by about 20-fold [8]. In a human pilot trial, supplementation with 250 mg or 500 mg QuerceFit® quercetin phytosome enhanced antioxidant capacity and improved upper respiratory function after 30 days [9]. To learn how phytosomes enhance absorption, check out this short lecture.
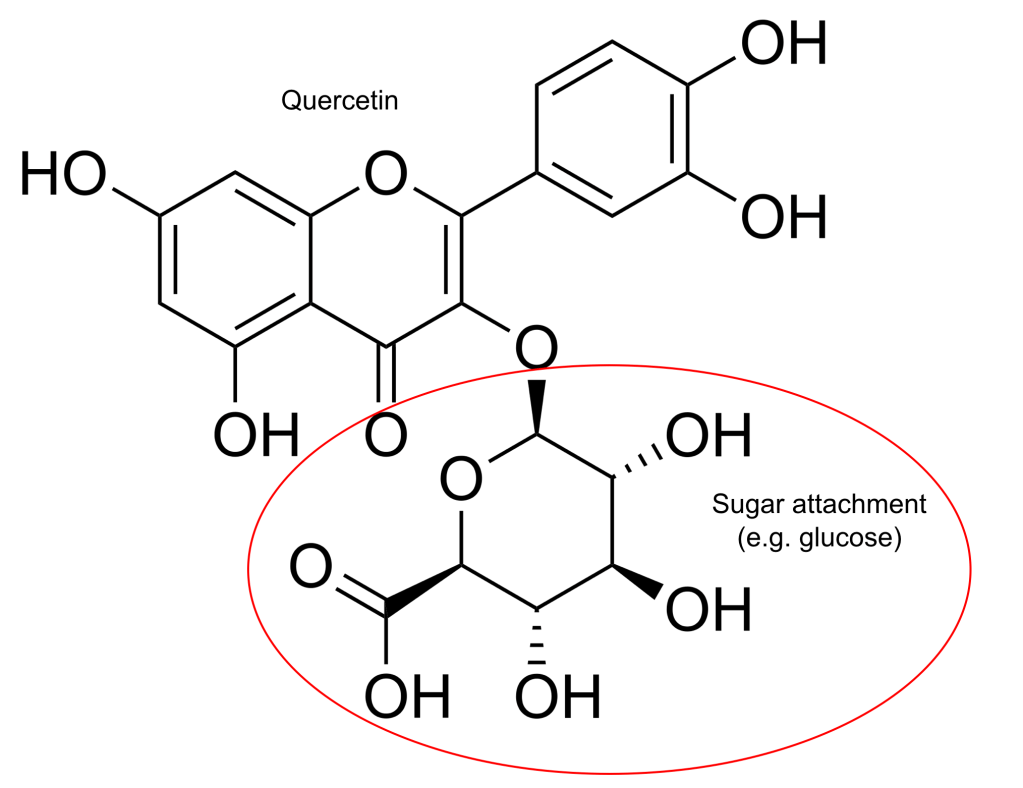
Another strategy to improve flavonoid absorption is glucose conjugation. A glucose moiety is added at the 3 and/or 4’ positions, greatly enhancing solubility. The resulting analog (isoquercetin), exhibits better bioavailability than its unconjugated counterpart [10-14]. Adding glucose makes passive diffusion impossible, but it opens a new door. Glucose enables the exploitation of a membrane glucose transporter. The glucose acts like forged passport, sending quercetin through the same carrier that takes up glucose after meals. This carrier-mediated route not only enables better absorption, but faster absorption. While non-glycosylated quercetin (aglycone) reaches peak plasma levels within 2-4 hours of an oral dose, isoquercetin achieves peak levels within 1 hour [13]. Plants, such as onions, make and store quercetin as isoquercetin. Absorption of isoquercetin from onions was 52% compared to 24% from a standard non-conjugated quercetin preparation you’d find in a typical supplement. Collectively, the studies show that isoquercetin may be 2-6-fold more bioavailable than the quercetin aglycone found in the vast majority of dietary supplements [10,13]
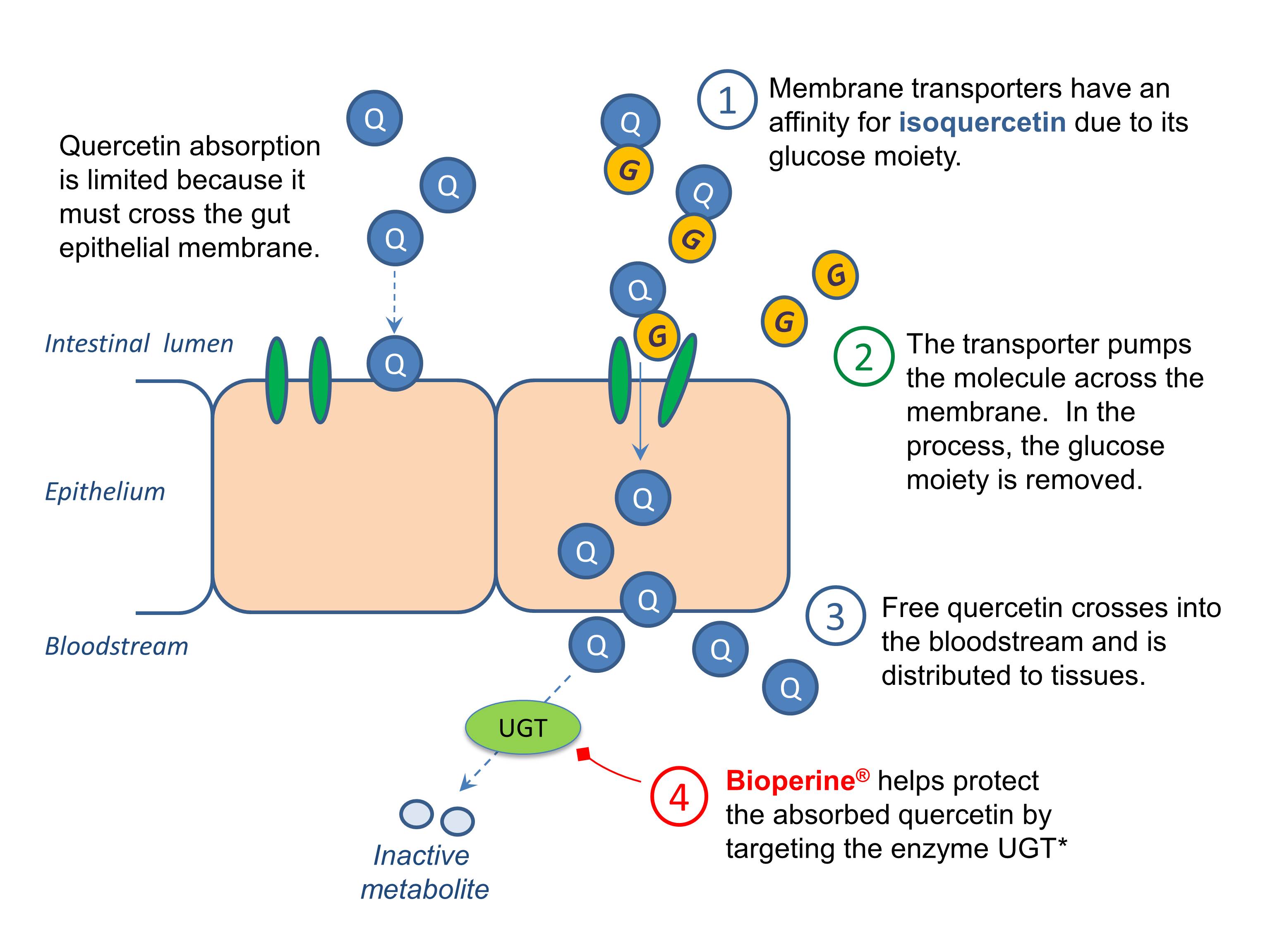
Solving for #3: Rapid presystemic metabolism
Whenever you enhance and/or accelerate absorption, you increase the potential to saturate, and thereby temporarily disable, phase II glucuronidation enzymes that normally inactivate quercetin and promote its elimination. Disabling these phase II enzymes solves for problem #3. But it’s not clear whether this really happens with phytosomes or isoquercetin.
Piperine is an alkaloid from black pepper that inhibits glucuronidation of polyphenols whose bioavailability is limited by phenolic hydroxyl groups (e.g. curcumin, quercetin, resveratrol) [16-18]. Piperine increased the bioavailability of curcumin in both rats and humans [15]. More recently, piperine enhanced the bioavailability of resveratrol [16] While more research is necessary to evaluate the effects of piperine on quercetin action in humans, preliminary evidence, published in July 2013, indicates that piperine can enhance the neurocognitive efficacy of quercetin in mice [18]. Since piperine may theoretically inhibit the metabolism and transport of various drugs, it should be used cautiously in individuals taking medications.
References
- Bischoff SC. Quercetin: potentials in the prevention and therapy of disease. Curr Opin Clin Nutr Metab Care (2008) 11(6):733-740.
- Heim K, Tagliaferro AR, Bobilya DJ. Flavonoid antioxidants: chemistry, metabolism and structure-activity relationships. J Nutr Biochem (2002) 13(10):572-584.
- Mlcek J, Jurikova T, Skrovankova S, Sochor J. Quercetin and Its Anti-Allergic Immune Response. Molecules. 2016 May 12;21(5):623. Click here for full text article
- Weng Z, et al. Quercetin Is More Effective than Cromolyn in Blocking Human Mast Cell Cytokine Release and Inhibits Contact Dermatitis and Photosensitivity in Humans. PLoS One. 2012;7(3):e33805. Click here for full text article
- Park HH, et al. Flavonoids inhibit histamine release and expression of proinflammatory cytokines in mast cells. Arch Pharm Res. 2008 Oct;31(10):1303-11.
- Ueno I, Nakano N, Hirono I. Metabolic fate of [14C] quercetin in the ACI rat. Jpn J Exp Med (1983) 53(1):41-50.
- Hollman PC, van Trijp JM, Mengelers MJ, et al. Bioavailability of the dietary antioxidant flavonol quercetin in man. Cancer Lett(1997) 114(1-2):139-140.
- Riva A, et al. Eur J Drug Metab Pharmacokinet. 2019; 44(2): 169–177.
- Cesarone MR, et al. Minerva Med. 2019 Dec;110(6):524-529.
- Hollman PC, van Trijp JM, Buysman MN, et al. Relative bioavailability of the antioxidant flavonoid quercetin from various foods in man. FEBS Lett (1997) 418(1-2):152-156.
- Manach C, Morand C, Demigné C, et al. Bioavailability of rutin and quercetin in rats. FEBS Lett (1997) 409(1):12-16.
- Gee JM, DuPont MS, Day AJ, et al. Intestinal transport of quercetin glycosides in rats involves both deglycosylation and interaction with the hexose transport pathway. J Nutr (2000) 130(11):2765-2771.
- Olthof MR, Hollman PC, Vree TB, Katan MB. Bioavailabilities of quercetin-3-glucoside and quercetin-4′-glucoside do not differ in humans. J Nutr (2000) 130(5):1200-1203.
- Erlund I, Kosonen T, Alfthan G, et al. Pharmacokinetics of quercetin from quercetin aglycone and rutin in healthy volunteers. Eur J Clin Pharmacol (2000) 56(8):545-553.
- Shoba G, Joy D, Joseph T, et al. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med (1998) 64(4):353-356.
- Johnson JJ, Nihal M, Siddiqui IA, et al. Enhancing the bioavailability of resveratrol by combining it with piperine. Mol Nutr Food Res (2011) 55(8):1169-1176.
- Srinivasan K. Black pepper and its pungent principle-piperine: a review of diverse physiological effects. Crit Rev Food Sci Nutr(2007) 47(8):735-48.
- Rinwa P, Kumar A. Quercetin along with piperine prevents cognitive dysfunction, oxidative stress and neuro-inflammation associated with mouse model of chronic unpredictable stress. Arch Pharm Res (2013) Jul 16. Advance Online Publication. DOI 10.1007/s12272-013-0205-4.
- Williams JA, Hyland R, Jones BC, et al. Drug-drug interactions for UDP-glucuronosyltransferase substrates: a pharmacokinetic explanation for typically observed low exposure (AUCi/AUC) ratios. Drug Metab Dispos. 2004 Nov;32(11):1201-8.
- Lee SH, Kim HY, Back SY, Han HK. Piperine-mediated drug interactions and formulation strategy for piperine: recent advances and future perspectives. Expert Opin Drug Metab Toxicol. 2018 Jan;14(1):43-57.

