Caloric restriction (CR) has attracted considerable attention in recent years as a method of prolonging lifespan and healthspan (duration of optimal health over the lifespan) in model organisms. CR enlists metabolic adaptations and renewal processes that improve cellular health and metabolic control. A prominent mechanism of CR is the induction of autophagy—the process of dismantling and clearing out damaged cellular components so that newer, healthier ones can take their place. Supporting this renewal process promotes healthspan and longevity in diverse models, from yeast to mammals [1,2].
In practice, caloric restriction is difficult to deploy because of low patient compliance and adverse effects. Alternative protocols involving alternate day fasting (ADF) may offer similar health benefits with better long-term adherence [3]. Another emerging strategy involves the use of CR mimetics (CRMs)—dietary or pharmaceutical compounds that may partially recapitulate the cellular effects of CR in lieu of dietary modifications.
CRMs lacked a unified definition until 2014, when Madeo and colleagues defined them as substances that inhibit protein acetylation [2]. Acetylation is a metabolic reaction that goes into overdrive when we consume lots of carbohydrates and fats –precursors of acetyl CoA, the body’s major source of acetyl groups. Acetylation activates proteins that block autophagy and mitophagy (autophagic disposal of damaged or unwanted mitochondria). Maintaining a low-calorie intake, and by extension, a “low acetylation state” relieves food-induced blockade of anti-aging enzymatic, epigenetic and transcriptional programs. In turn, CR-induced hypoacetylation “tunes up” metabolism, insulin sensitivity, mitochondrial renewal and other predicates of healthy aging [2,4].
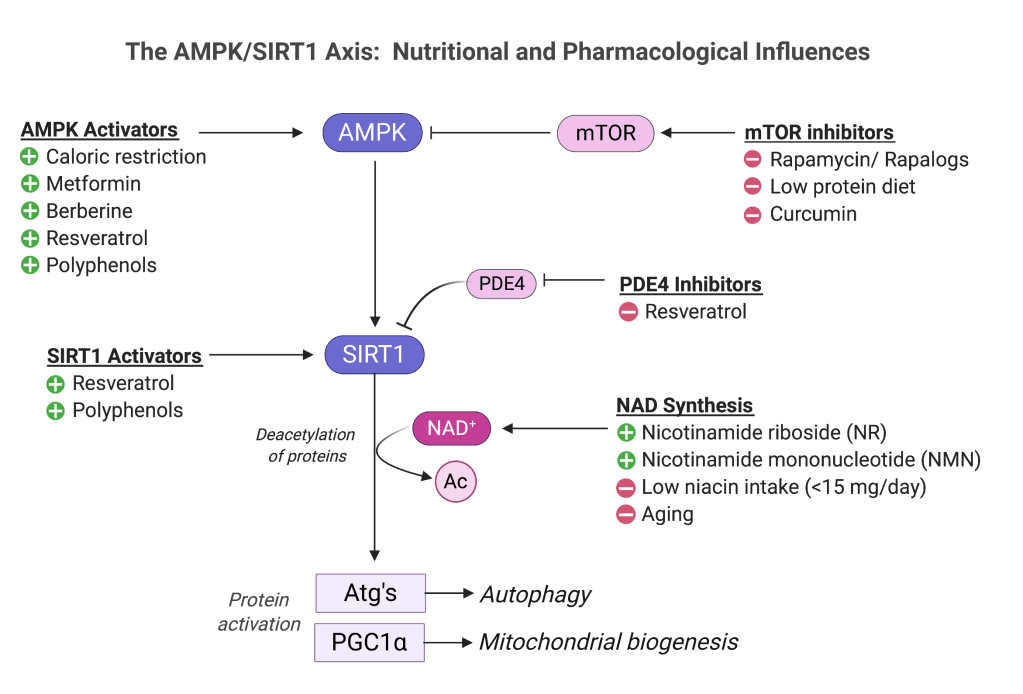
While CRMs cannot deplete acetyl groups the way fasting does, they can directly interact with enzymes that stick the acetyl groups onto proteins (acetyltransferases) or clip them off (deacetylases) [2,4]. The most famous example is sirtuin 1 (SIRT1), an NAD-dependent enzyme that mediates the major benefits of CR [5,6]. SIRT1 is potently stimulated by an upstream energy sensor called AMP kinase (AMPK). AMPK detects a drop in ATP (resulting from fasting or exercise), enlisting SIRT1 to activate autophagy drivers called Atg’s (Figure 1).
A long list of natural and synthetic compounds may complement CR by interacting with the AMPK/SIRT1 axis at various points. Here are a few examples:
- Resveratrol may allosterically activate the SIRT1 protein and/or act indirectly, i.e. by AMPK activation [2,7].
- Rapamycin, a macrolide antibiotic, supports AMPK/SIRT1 by blocking the mammalian target of rapamycin (mTOR) pathway. Rapamycin is the antecedent for an emerging class of experimental anti-aging pharmaceuticals known as “rapalogs” [8]. High protein intake activates mTOR, so reducing protein intake could support healthy aging. However, the anabolic effects of mTOR are instrumental in preserving lean body mass, so adequate protein should be maintained.
- Nicotinamide riboside and nicotinamide mononucleotide (NMN) activate SIRT1 by elevating levels of its co-substrate, NAD+ [2,9].
- Metformin, a biguanide with complex metabolic effects, stimulates AMPK by reducing ATP levels, mimicking energy restriction [10].

For a deeper dive into CR, its mechanisms, and interactions with nutritional compounds, here are two excellent full-text reviews:
- Dai et al. Sirtuin activators and inhibitors: Promises, achievements, and challenges. Pharmacol Ther 2018;188:140-154.
- Burkewitz et al. AMPK at the nexus of energetics and aging. Cell Metab 2014; 1;20:10-25.
Summary & Key Points
- Caloric restriction, a promising strategy for healthy aging, works (in part) through the stimulation of autophagy [2,4,9,10].
- CR modifies the function of various proteins involved in metabolism and autophagy by changing their acetylation state [2,4].
- SIRT1 drives CR-induced autophagy by activating autophagy-inducing proteins and other effectors via NAD+-dependent acetylation [4-6]. NAD+ is made from dietary niacin, and its biosynthesis declines with age [9].
- SIRT1 and its regulators respond to various natural and synthetic compounds, some of which partially mimic the effects of caloric restriction at the cellular level. These compounds are called caloric restriction mimetics (CRMs) [2,4,9].
References
- Nakamura S, Yoshimori T. Autophagy and Longevity. Mol Cells. 2018 Jan 31;41(1):65-72.
- Madeo F, Pietrocola F, Eisenberg T, Kroemer G. Caloric restriction mimetics: towards a molecular definition. Nat Rev Drug Discov. 2014 Oct;13(10):727-40.
- Stekovic S, Hofer SJ, Tripolt N, et al. Alternate Day Fasting Improves Physiological and Molecular Markers of Aging in Healthy, Non-obese Humans. Cell Metabolism, 2019; DOI: 10.1016/j.cmet.2019.07.016.
- Madeo F, Carmona-Gutierrez D, Hofer SJ, Kroemer G. Caloric Restriction Mimetics against Age-Associated Disease: Targets, Mechanisms, and Therapeutic Potential. Cell Metab. 2019 Mar 5;29(3):592-610.
- Lee, I. H. et al. A role for the NAD-dependent deacetylase SIRT1 in the regulation of autophagy. Proc Natl Acad Sci. 105, 3374 (2008).
- Mercken EM et al. SIRT1 but not its increased expression is essential for lifespan extension in caloric-restricted mice. Aging Cell 13, 193–196 (2014).
- Park, S.-J. et al. Resveratrol ameliorates aging-related metabolic phenotypes by inhibiting cAMP phosphodiesterases. Cell 148, 421–433 (2012).
- Schreiber, K.H., Arriola Apelo, S.I., Yu, D. et al. A novel rapamycin analog is highly selective for mTORC1 in vivo. Nat Commun 10, 3194 (2019).
- Dai H, Sinclair DA, Ellis JL, Steegborn C. Sirtuin activators and inhibitors: Promises, achievements, and challenges. Pharmacol Ther. 2018 Aug;188:140-154.
- Burkewitz K, Zhang Y, Mair WB. AMPK at the nexus of energetics and aging. Cell Metab. 2014 Jul 1;20(1):10-25.

