The endocannabinoid system (ECS) plays critical roles in regulating diverse biological processes, ranging from pain sensitivity to immune function. Despite its widespread acclaim and proven medical relevance, everything we know about this physiological system was discovered fairly recently.
It all began with simple questions about an ancient medicinal plant. Cannabis sativa is an herbaceous flowering spceies indigenous to eastern Asia, with records of therapeutic and recreational use dating back more than 45 centuries.
A brief history
While the clinical effects of cannabis on basic body functions were well known, the chemical constituent responsible for its actions remained a mystery until 1964, when Israeli chemist Raphael Mechoulam identified and isolated delta-9 tetrahydrocannabinol (THC).

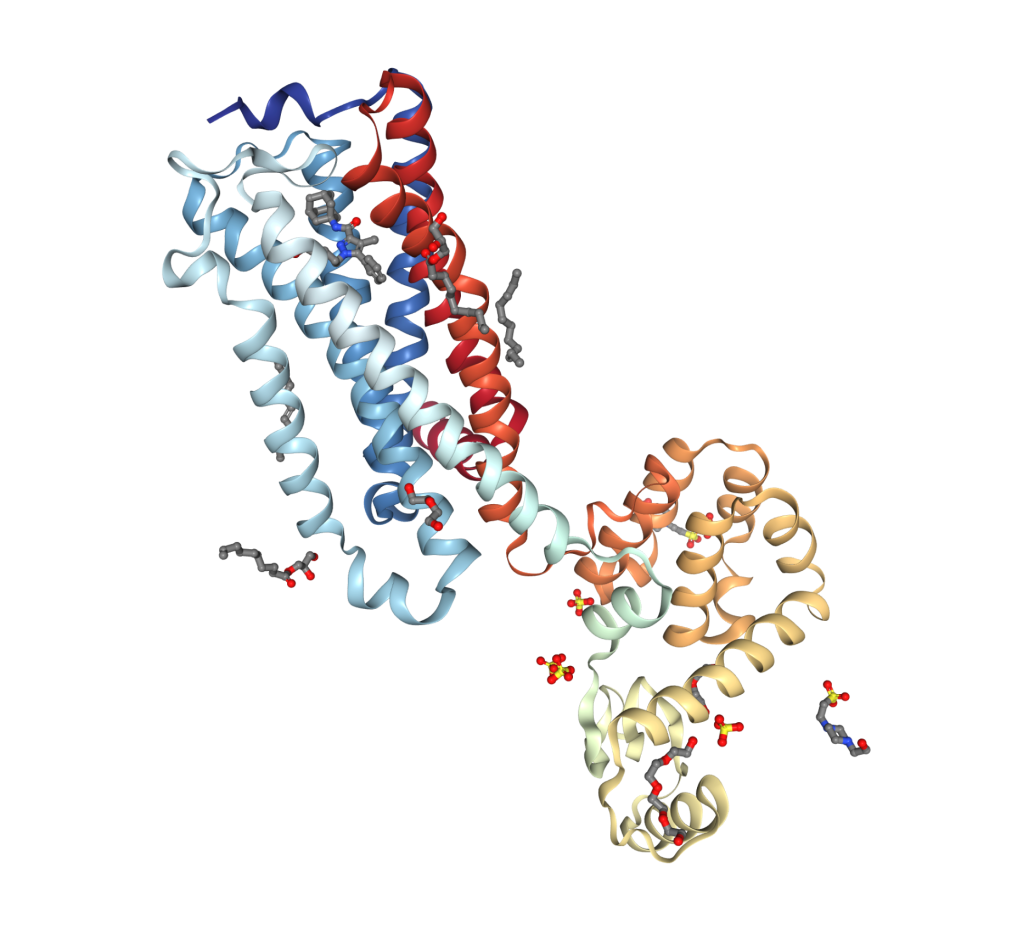
Further exploration of how THC and other phytocannnabionids worked were hindered by the limited technologies available to molecular biologists and chemists at the time. It wasn’t until 1988 when THC was found to bind to a receptor, known as CB1, which is abundantly expressed in the brains of vertebrates, particularly in regions responsible for cognition, emotions, appetite, and motor coordination.
While THC’s affinity for central CB1 receptors explained its psychotropic and euphoriant effects, it could not explain the many peripheral actions of the drug. In 1993, researchers discovered a second cannabinoid receptor subtype known as CB2, which was later detected in cells of the immune system, gut and other peripheral tissues.
Homeostasis: The elusive raison d’être
As scientists were solving these old mysteries about the molecular pharmacology of cannabis, a new and bigger question was emerging: What physiological purpose do these receptors serve? Nature wouldn’t conserve them unless they performed a natural physiological function. Scientists began searching for endogenous ligands, which soon culminated in the discovery of two endocannabiniods: anandamide (AEA) and 2-arachidonoylglycerol (2-AG) in the mid 1990s.
The purpose of the ECS is to aid in supporting homeostasis throughout the body. In doing so, it ensures that cells communicate effectively but not excessively, and steps in when things get out of balance.
The ECS is comprised of 3 components:
Endocannabinoids, which are derived from membrane lipids. Anandamide and 2-arachidonoylglycerol are the important examples.
Receptors, which include, but are not limited to, CB1 and CB2. Both are expressed diffusely, but CB1 predominates in the CNS.
Enzymes that control the synthesis and degradation of endocannabinoids. The ECS is largely under enzymatic control and the levels of anandamide and 2-AG present at any given time are determined by the balance of enzyme-mediated synthesis and degradation.
How it works
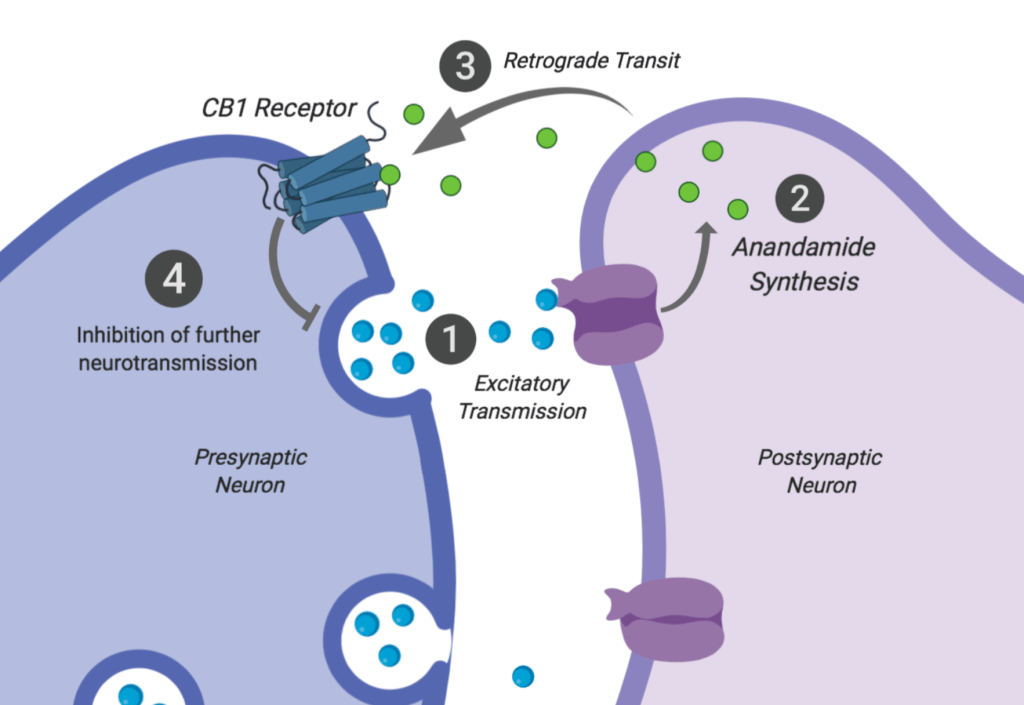
The ECS keeps excessive signaling at bay by inhibiting further release of the inciting stimulus. For example, in the immune system, it inhibits the release of pro inflammatory cytokines. In the CNS, it inhibits certain types of neurotransmission. When excitatory neurotransmitters activate their receptors on the postsynaptic neuron, the resulting depolarization triggers the synthesis of anandamide, which travels in a retrograde pattern to the presynaptic cell. It then binds to the CB1 receptor, G-protein-coupled receptor that increases intracellular ion concentrations to inhibit further degranulation of the excitatory substance.
The definition of the ECS remains vague
It is important to recognize that endocannabinoids interact with many other molecular targets beyond CB1 and CB2. For example, anandamide binds, activates and desensitizes an ion channel on certain neurons known as TRPV1 (transient potential vanilloid type 1, formerly known as the vanilloid receptor type 1) to reduce the perception of pain. Endocannabinoids also modulate other types of receptors that have nothing to do with pain, anxiety, sleep or other commonly presumed indications for cannabinoids.
While the ECS is a highly attractive therapeutic target for a variety of conditions, its diverse receptor and enzyme constituents appropriately preclude designating something strictly as an ECS modulator. ECS modulating drugs are likely to belong to other drug classes (e.g. corticosteroids, SSRIs, PPAR agonists) or lifestyle interventions (e.g. exercise, meditation) with proven actions against more relevant, non ECS effectors. Any newly recognized substance that allegedly works by modulating the ECS, or acclaimed in similar parlance, will present its own unique clinical indications and therapeutic profile.
With a healthy dose of skepticism, treat all emerging ECS modulators differently, with careful consideration of supporting data and evaluation of specific clinical indications.

