Hemp is a complex natural product with hundreds of constituents including over 100 different phytocannabinoids, most of which still await pharmacological interrogation. The most abundant and well-researched non-psychotropic phytocannabinoid is known as cannabidiol, or CBD. Regarding its pharmacodynamics and molecular targets, many questions persist.
Unlike THC, CBD fails to activate CB receptors directly. This is partly due to the differential recognition of bent vs. planar conformation of CBD and THC, respectively, at the receptor ligand binding sites. However, CBD may potentiate anandamide-mediated activation of CB receptors by attenuating anandamide hydrolysis. This is achieved by weak inhibition of fatty acid amide hydrolase (FAAH), the enzyme that degrades anandamide.
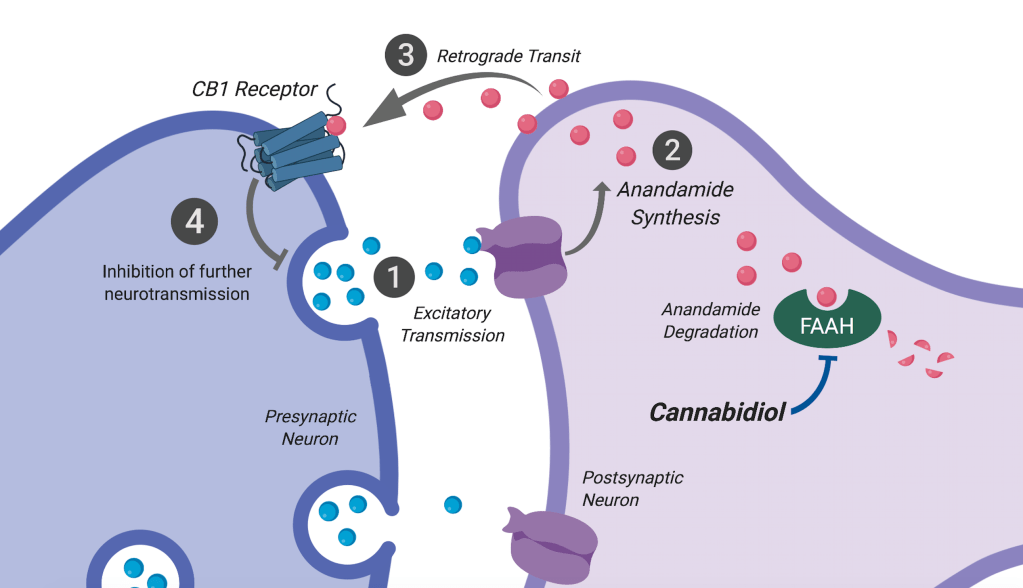
So, how does CBD work?
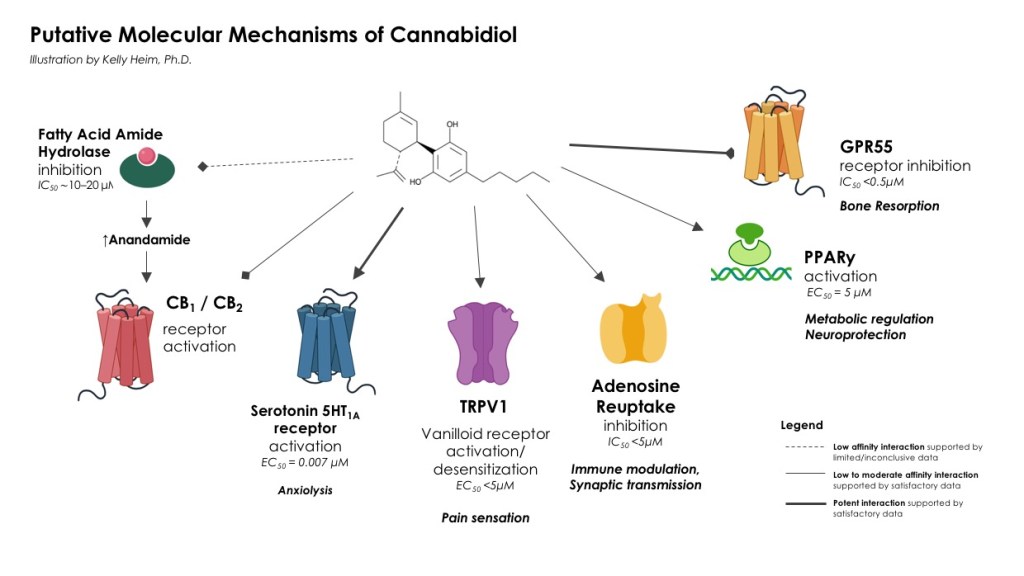
Until further research explores how CBD works in humans receiving the low doses (<30 mg) widely available as supplements, we don’t really know how it works. With confidence, I can say there is very little, if any, involvement of direct interaction with canonical CB receptors. Instead, evolving research underscores disparate effectors such as serotonin receptors, ion channels and reuptake systems.
The following mechanisms are well-supported by in vitro and animal data [1-13]:
- Activation of serotonin 5HT1A receptors plays a part in the anxiolytic effects of CBD in the limbic system, a brain region involved in emotional processing, mood and behavior.
- Inhibition of transient receptor potential cation channel subfamily V member 1 (TRPV1) ion channels. In the peripheral nervous system, CBD modulates neurosensory responses by activating and subsequently desensitizing them.
- Adenosine receptor activation and reuptake inhibition. Adenosine is an inhibitory neurotransmitter and immune modulator.
- Activation of peroxisome proliferator activated receptor gamma (PPARγ), which is involved in metabolic regulation and immune function.
- Blockade of GPR55, a novel interaction implicated in osteoclast function.
References
- Peres FF, Lima AC, Hallak JEC, et al. Front Pharmacol. 2018 May 11;9:482.
- Bisogno T, Hanus L, De Petrocellis L, et al. Br J Pharmacol. 2001;134:845–85
- Leweke FM, Piomelli D, Pahlisch F, et al. Transl Psychiatry. 2012;2:e94.
- De Petrocellis L, Ligresti A, Moriello AS, et al. Br J Pharmacol. 2011;163:1479–1494.
- Leishman E, Manchanda M, Thelen R, et al. Cannabis Cannabinoid Res. 2018 Nov 30;3(1):228-241.
- Espejo-Porras, F., Fernandez-Ruiz, J., Pertwee, R. G., Mechoulam, R., and Garcia, C. (2013). Neuropharmacology 75, 155–163.
- Gomes, F. V., Del Bel, E. A., and Guimaraes, F. S. (2013). Prog. Neuropsychopharmacol. Biol. Psychiatry 46, 43–47.
- Pazos, M. R., Mohammed, N., Lafuente, H., et al. (2013). Neuropharmacology 71, 282–291.
- Hind, W. H., England, T. J., and O’Sullivan, S. E. (2016). Br. J. Pharmacol. 173, 815–825.
- Sartim, A. G., Guimaraes, F. S., and Joca, S. R. (2016). Behav. Brain Res. 303, 218–227.
- Lee, J. L. C., Bertoglio, L. J., Guimaraes, F. S., and Stevenson, C. W. (2017). Br. J. Pharmacol. 174, 3242–3256.
- Costa et al. Br J Pharmacol. 2004 Sep; 143(2): 247–250.
- Bisogno T, Hanus l, de Petrocellis l, et al. Br. J. Pharmacol. 2001;134:845–852.

